Completed Winship Clinical Trial for Glioblastoma Patients
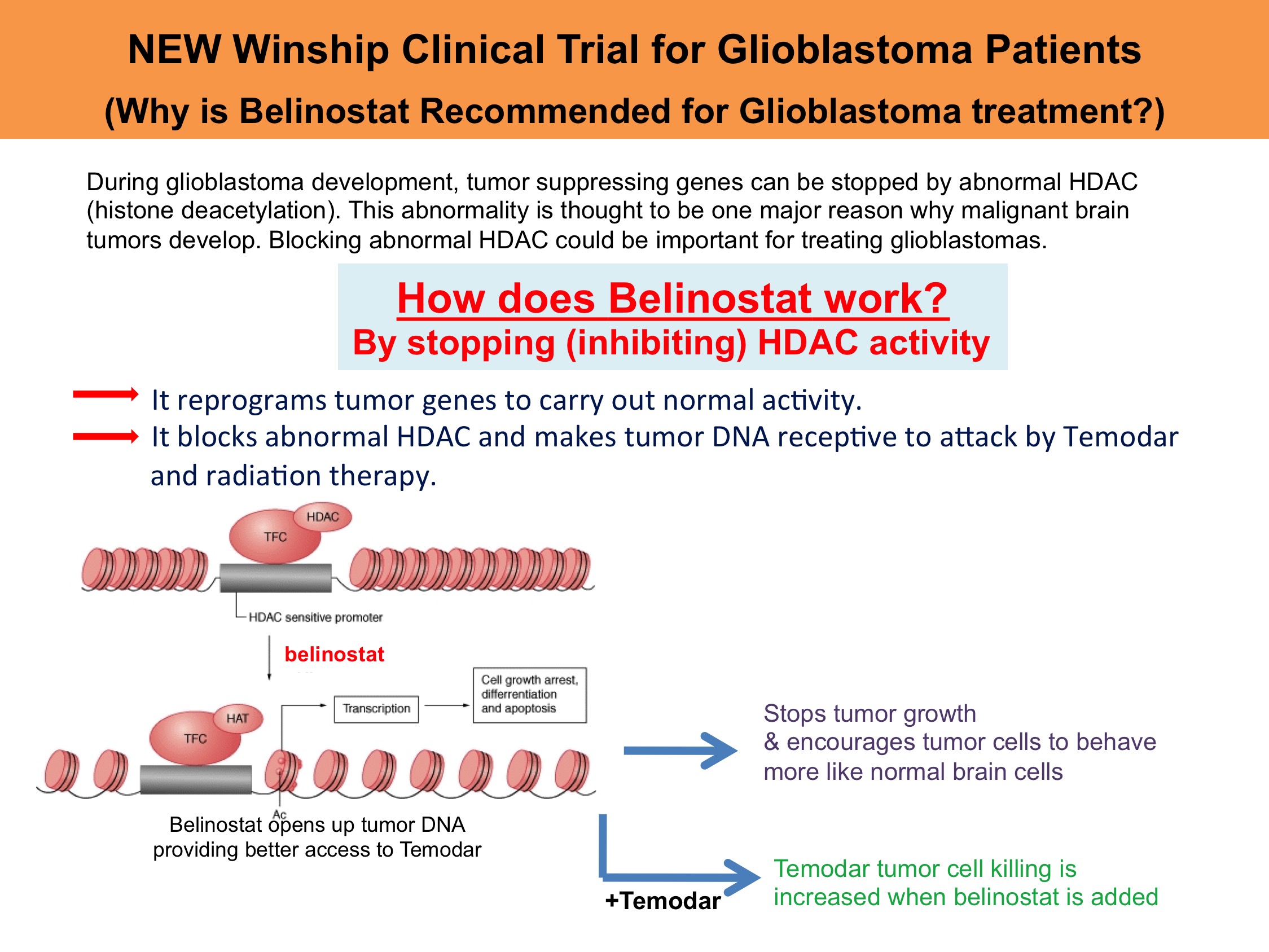
A completed clinical trial at Emory’s Winship Cancer Institute aimed to improve the effectiveness of chemoradiation therapy in brain cancer patients by adding a new class of drug to the current standard of care while obtaining a new form of advanced MRI called spectroscopic MR imaging that may help predict who will respond to this therapy. Belinostat could be important for treating glioblastomas because of its two main mechanisms of action: forcing tumor cells to re-differentiate and sensitizing tumors to temodar and radiation therapy. Forcing tumor cells to re-differentiate essentially reprograms tumor genes to stop tumor growth and to carry out normal brain cell activity. Sensitizing tumors to chemoradiation is significant because it increases the efficacy of the current standard of care for brain cancer patients. Thirty-six other registered clinical trials have used or are using belinostat, but this is the first trial that tests its use in brain cancer. The use of this advanced spectroscopic MR imaging to measure the metabolite content of infiltrating brain tumor is a unique aspect of this trial that may eventually allow us to determine at an early time who will benefit from a therapy like Belinostat.
Clinical Principal Investigators:
Hui-Kuo Shu, MD, PhD, Radiation Oncologist at Emory Winship Cancer Institute
Jeffrey Olson, MD, Neurosurgeon at Emory Winship Cancer Institute